So, picture this: you’re at a family gathering, and your aunt starts telling you about her latest bout with a stubborn infection. She’s throwing around words like “penicillin” and “cephalosporins,” and you’re just standing there, nodding along, trying to catch up. It’s like she’s speaking another language!
But seriously, antibiotic pharmacology is way cooler than it sounds. It’s all about how these little miracle workers—yeah, the antibiotics—interact with our bodies to fight infections. Imagine tiny soldiers going to battle against bacteria while you sip your coffee.
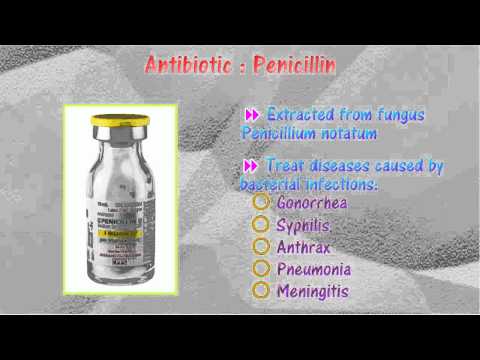
And here’s a wild fact: did you know that the discovery of penicillin back in the 1920s basically kicked off the antibiotic revolution? Before that, an infected cut could be a real game changer for anyone!
Anyway, in this chat about antibiotics, we’re gonna dig into what they are, how they work their magic in modern medicine, and why they matter so much today. So grab a snack and let’s get into it!
Comprehensive Guide to the Classification of Antibiotics: Downloadable PDF Resource for Scientific Study
The classification of antibiotics can be a bit overwhelming at first, but once you break it down into chunks, it’s much easier to digest. So let’s chat about it!
Antibiotics are basically drugs that fight bacterial infections. They work by either killing the bacteria or inhibiting their growth. However, not all antibiotics are created equal—there’s quite a variety out there, and they all fit into different categories based on how they work and what they’re used for.
First off, antibiotics can be classified based on their chemical structure. This is like categorizing them by family traits. Here are some common groups:
- Beta-lactams: Think of penicillin and its cousins like amoxicillin. They have a special ring in their structure that makes them super effective against many bacteria.
- Tetracyclines: These guys, like doxycycline, are broad-spectrum antibiotics. They can tackle a wide variety of bacteria.
- Aminoglycosides: This group includes drugs like gentamicin that are often used for serious infections caused by gram-negative bacteria.
- Macrolides: Examples include erythromycin and azithromycin; they’re great for treating respiratory infections.
On the other hand, you have classification based on how they affect bacteria. Some antibiotics attack the walls of the bacteria (like beta-lactams), while others target protein synthesis (like tetracyclines).
Then we have broad-spectrum vs narrow-spectrum antibiotics. Broad-spectrum ones act against a wide range of bacteria. Narrow-spectrum antibiotics only target specific types. The choice between these two can make a huge difference in treatment—you know? Using broad-spectrum when it’s not necessary might lead to antibiotic resistance!
And speaking of resistance, here’s where it gets real: overusing or misusing these medications can cause bacteria to adapt over time so they don’t respond to the drug anymore. This is why doctors usually try to prescribe only what’s necessary.
A little story: I remember visiting my grandma when she was sick with an infection. The doctor gave her an antibiotic that worked wonders without messing up her whole system—no side effects! That’s because he chose the right one for her specific infection.
Lastly, there’s ongoing research in this field; scientists keep discovering new compounds and refining classifications as they learn more about how these drugs interact with our bodies and bacterial pathogens.
So remember—antibiotics are essential tools in modern medicine but come with responsibilities. Understanding how they’re classified helps us use them wisely!
Comprehensive Analysis of Antimicrobial Resistance: Downloadable Research Paper PDF for Scientific Study
Antimicrobial resistance is one of those topics that might sound a bit, you know, overwhelming at first. But don’t worry—we’ll break it down together! So, antimicrobial resistance basically happens when bacteria, viruses, fungi, or parasites evolve and become resistant to medications that once killed them or inhibited their growth. This is a major problem today and affects how we treat infections.
What’s the big deal? Well, let me tell you a little story. A few years back, my friend got an infection after a surgery. Doctors prescribed antibiotics right away because that’s usually how these things go. But guess what? The bacteria causing the infection was resistant to those antibiotics! In the end, they had to use a really strong medication that had crazy side effects just to tackle it. It was scary—like something out of a medical drama.
So now let’s get into some key points about this whole antimicrobial resistance situation:
- Causes: Overuse and misuse of antibiotics are huge factors. Seriously, people often take them for viral infections like the common cold when they don’t work at all against viruses.
- Consequences: When treatments stop working, simple infections can turn deadly. We might be heading back to the pre-antibiotic era if this keeps up—yikes!
- Research efforts: Scientists worldwide are hustling to understand and combat this issue. They’re looking for new drugs and alternative therapies while also digging into how resistance happens at a genetic level.
- Prevention: One way to tackle this problem is through education on responsible antibiotic use. Like knowing when it’s truly necessary to take them can make a world of difference!
Antibiotic pharmacology plays a massive role in modern medicine by targeting bacterial infections effectively when used appropriately. On the flip side, though, inappropriate use leads to resistance.
It’s not all doom and gloom! There are initiatives aiming at reducing antibiotic misuse in healthcare settings as well as in agriculture where they’re often used preventively in livestock farming (which totally adds to the problem). And there are efforts to develop vaccines that could help prevent some bacterial infections altogether.
Talking about research papers on this topic—yeah, they can get pretty dense and academic—but they’re super important for scientists digging deeper into understanding resistance mechanisms or finding new drugs. If you’re keen on checking out some downloadable research papers or PDFs related to Antibiotic Pharmacology and Antimicrobial Resistance , tons of universities have resources available online where you can access these studies.
Ultimately, what we need is action— from individuals taking care with antibiotics at home to policymakers making decisions about regulatory measures in healthcare settings. It’s all connected!
So remember: next time you feel under the weather and think about grabbing antibiotics after chatting with your doctor—just give it some thought! You want those little bacteria fighting fit so they don’t come back stronger than ever later on.
Understanding Antibiotics: Definition, Classification, and Their Role in Science
Antibiotics are pretty amazing, honestly. They’re like tiny warriors fighting off bacteria that make us sick. Basically, they’re substances that can kill or inhibit the growth of bacteria. The cool part? They don’t really mess with our own cells—just the bad guys.
Now, let’s break this down a bit. You can think of antibiotics in a few different ways based on how they work and what types of infections they target.
- Bactericidal: These guys go all out and kill bacteria directly. Like, boom! An example here would be penicillin.
- Bacteriostatic: Instead of killing, they just slow down bacterial growth so your immune system can do its thing. Tetracycline is a classic example.
When it comes to classes of antibiotics, there are some big players you might want to know about:
- Penicillins: This group is one of the earliest antibiotics discovered and still widely used today!
- Cephalosporins: Kind of like the cousins to penicillins but with a broader spectrum.
- Aminoglycosides: These are usually for more serious infections and work well against gram-negative bacteria.
- Macrolides: Great for respiratory infections; erythromycin is a common name here.
- Fluoroquinolones: Think ciprofloxacin and levofloxacin—they’re often used for urinary tract infections.
So, why do we care about these little warriors? Well, antibiotics have transformed modern medicine! Before them, even simple bacterial infections could lead to serious complications or even death. Imagine having an infected cut during World War II—yikes! A simple shot of penicillin changed all that.
But it’s not all sunshine and rainbows. Over time, some bacteria have become resistant to certain antibiotics through misuse or overuse. It’s like they’ve learned how to dodge our bullets! So now scientists are working hard on research to find new antibiotics and develop strategies to combat resistance.
In the lab, researchers study how these drugs affect bacteria at a molecular level. It’s fascinating stuff! They examine everything from how quickly the antibiotic works to what happens when it encounters resistant strains.
Understanding antibiotics isn’t just about knowing their names; it’s also about grasping their impact on our health and society. We need them to control infections—but we have to use them wisely!
In summary, antibiotics are essential tools in modern medicine but come with challenges we need to address together as responsible users and stewards of public health! Feel free to reflect on your own experiences with them—you might find there’s more than meets the eye when it comes to those little pills we often take for granted.
Antibiotics, right? They’re like those heroes in our medicine cabinet. I mean, imagine a world without them. Seriously, think about it! A simple cut could lead to an infection that spirals out of control. But today, thanks to antibiotics, we’ve tilted the scales back in our favor. That’s super important for modern medicine.
So here’s the thing: antibiotics are not just about killing bacteria. It’s so much more than that. They work by targeting specific parts of bacterial cells—kinda like how a key fits into a lock. You’ve got penicillins and tetracyclines, each with their own style and method of action. Some mess with cell walls while others disrupt protein synthesis. Pretty neat how they outsmart those pesky bugs!
But here’s where it gets a little sticky—pun intended! Overusing or misusing antibiotics has made some bacteria more resilient. It’s like when you keep feeding your pet the same treat; eventually, they get used to it and won’t come running anymore! Antibiotic resistance is one of the biggest challenges in healthcare today. You might hear stories from doctors who face patients with infections that just won’t budge because the bacteria have learned how to evade treatment.
I remember hearing about a friend who had to battle an infection after surgery—you know, something routine but still a bit scary? The doctor told him they couldn’t give him the usual meds because his bacteria were resistant to them. Imagine feeling that helplessness! Thankfully, modern research is working hard on this front, discovering new antibiotics and even exploring alternatives like bacteriophage therapy or vaccines.
And let’s not forget about our gut microbiome—the friendly microbes living inside us that help with digestion and even influence our immune system! Antibiotics can sometimes wipe out these good guys along with the bad ones, leaving us vulnerable to other issues down the line.
It makes you think about balance too; we need these incredible drugs but also have to be smart about how we use them. So while antibiotic pharmacology continues advancing through research and clinical trials—like developing better-targeted treatments—we also have this responsibility to use them wisely.
In short, antibiotics are powerful allies in our fight against infections—but they’re not invincible superheroes without their faults! The journey in medicine is ongoing; every discovery opens new possibilities and challenges alike. And isn’t that what makes science so thrilling?