So, picture this: you’re blowing bubbles at a park. One moment they’re floating gracefully, and then—pop!—they burst into a million tiny sparkles. It’s like watching magic happening right before your eyes.
But here’s the kicker: those delicate little spheres are actually packed with fascinating science! Seriously, there’s so much going on in each bubble that you’d never guess just by looking.
You know how they float effortlessly, dancing in the wind? That’s all thanks to some cool physics at play. And believe it or not, bubble science isn’t just for kids having fun. It’s got some real-world applications too—like in medicine or engineering!
So let’s break it down and uncover why these soapy wonders are way more than just a childhood pastime. Curious? Let’s get into it!
Exploring the Physics of Bubbles: Understanding Surface Tension and Fluid Dynamics in Science
So, bubbles! Isn’t it cool how something so simple can be packed with science? When you blow a bubble, you get to see a little world of physics come to life right before your eyes. It’s all about surface tension and fluid dynamics. Let’s break it down.
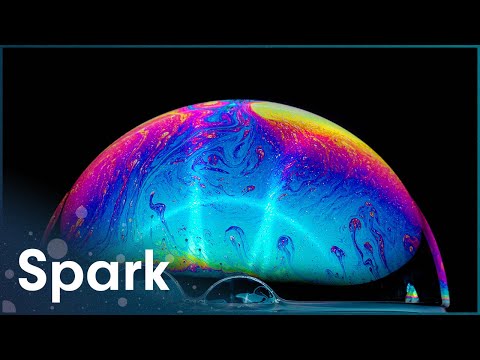
First off, **what is surface tension**? Picture a tight rubber band stretched over the surface of water. That’s kind of what happens on the surface of a liquid. Molecules at the surface stick together more strongly than those deep down. They form a sort of invisible skin that makes bubbles possible. When you blow air into soap solution, this skin stretches out and forms a bubble!
Now, let’s chat about **how bubbles are made**. You’ve probably seen how things like dish soap can create tons of bubbles when mixed with water. That’s because soap reduces the surface tension of water. With less tension pulling on those water molecules, they can expand more easily when you blow air into them. It’s like having an easier time stretching out your favorite pair of pants after eating a big meal—everything just relaxes.
But wait, there’s more! We should talk about **fluid dynamics** too. Essentially, this is how liquids move and behave under different conditions—like when air blows through the soap solution to make bubbles. The way air and liquid interact is super important in understanding why some bubbles float while others pop right away.
When you see huge soap bubbles floating delicately in the air, that’s not just luck—it’s physics at play! Bubbles form into spheres because this shape minimizes their surface area for the volume they enclose, which is basically nature’s way of being efficient.
Another cool point to think about is how gravity plays its role here too! If you were to make giant bubbles on Earth—let’s say the size of beach balls—they would behave differently than if they were made on the Moon due to lower gravitational pull there. Fun thought exercise, huh?
You might also find it interesting that scientists use bubbles in various applications. For instance, in medicine or environmental science, researchers explore tiny bubbles called microbubbles for drug delivery or removing pollutants from water!
And take my word for it: there’s nothing quite like witnessing your own homemade bubbles dance around in sunlight—each one sparkling with possibilities thanks to that magical world governed by physics!
So, yeah! The next time you’re blowing bubbles or even just observing them float by your window, remember there’s so much science swirling around inside those delicate spheres! It’s really amazing how much there is behind something so ordinary yet mesmerizing as a bubble.
Understanding Bubble Law in Physics: Principles and Applications in Scientific Research
So, bubble law in physics might sound a bit nerdy, but it’s actually super cool when you break it down. Basically, it’s all about understanding how bubbles form, grow, and eventually pop. Think of them as little floating mysteries that hold a lot of science inside.
Firstly, let’s chat about the principles. The law governing bubbles is tied to concepts like surface tension and pressure. You know when you blow soap into a bubble? That beautiful sphere forms because of surface tension—the skin of the bubble trying to minimize its surface area. It’s like an invisible force pulling things together! When you see a bubble floating, it’s keeping that shape because the pressure inside is balanced by the pressure outside—pretty neat, right?
Now, onto applications. Bubbles aren’t just for kids’ play; they have serious applications in scientific research. For example:
You might find this fascinating: there are even studies about how bubbles can influence climate change! Seriously! When ocean waves break, they create bubbles that release gases like CO2 into the atmosphere or trap heat in the water below.
On a personal note, I remember watching my younger sibling try to blow the biggest bubble possible during summer days at the park. Each pop was met with squeals of joy (or frustration!). But thinking back on it now, every time that bubble burst was actually an example of those physics principles we’re talking about—showing how everything is interconnected.
So next time you see a bubble floating by or watch one pop dramatically—you’ll know there’s way more going on than just fun! It’s all about those little laws of physics at play!
The Science Behind Unpoppable Bubbles: Exploring Physics and Material Science
So, let’s talk about bubbles. You know, those little spheres of joy that float around when you’re enjoying a sunny day outside? Well, there’s some serious science behind why they pop — or sometimes, why they don’t! It’s all about the physics and material science working together.
First off, a bubble is basically just a thin film of liquid surrounding a pocket of gas. When you blow into a bubble wand, air gets trapped inside this liquid film. But here’s where it gets tricky: the surface tension in that liquid wants to pull everything tight and keep the gas in. This is what gives bubbles their round shape! Surprising, right?
Now let’s get into the reasons why some bubbles can be unpoppable—like those amazing giant ones you see at festivals or on YouTube. These bubbles are often made using special solutions that include ingredients like glycerin or dawn dish soap.
Why these ingredients? It all comes down to how they interact with water molecules. Here’s the deal:
- Glycerin: This sticky stuff slows down evaporation. So instead of popping as quickly as regular soap bubbles do, glycerin helps them last longer.
- Soap molecules: They have hydrophilic (water-attracting) and hydrophobic (water-repelling) parts. This dual nature helps to create that strong interface necessary for keeping gas trapped inside.
- Additives: Sometimes sugar is added too! Sugar increases viscosity, which can make those bubbles even tougher.
You might have seen videos of people using giant wands or even homemade bubble solutions creating huge and seemingly unpoppable bubbles. It’s pretty cool and also has to do with how pressure works! When you increase the size of the bubble without increasing its thickness enough, it creates an enormous amount of stress on the surface tension at play.
Oh, and let me tell you about something super interesting: when a bubble is floating in the air, it’s actually experiencing forces from all kinds of angles—the wind can push against it while gravity pulls it down. The balance between these forces determines if it will stay afloat or pop! Like that time I tried to catch one with my palm—but instead ended up splashing soapy water everywhere! Yeah… not my finest moment!
In terms of applications beyond just playtime fun, understanding bubble physics has real-world impacts too! Industries utilize this knowledge for things like improving foam insulation materials or making better aerated food products like whipped cream.
So there you have it—the science behind unpoppable bubbles isn’t just about having fun; it’s also rooted in some seriously cool physics and material science concepts that teach us more about our world! Who knew something so simple could be so complex?
You know those moments when you blow bubbles and just stare at them, mesmerized? They float, shimmer, and pop in a split second. But there’s actually a whole world of physics behind those little orbs that makes them so captivating. Seriously, it’s like magic!
So here’s the deal: bubbles are basically thin films of liquid surrounding a gas. Most often it’s air trapped inside a layer of soap and water. That soap part is key, you see? It lowers the surface tension of the water, which allows bubbles to form larger than they could otherwise. Ever tried blowing one from just plain water? Yeah, it doesn’t work quite as well.
I remember one summer day as a kid when my friend and I decided to have an epic bubble-blowing contest. We mixed dish soap with some water in an old bucket and ended up with not only big bubbles but also tiny ones that stuck to each other like they were having their own party! It was such a joy seeing how they danced in the sunlight; their colors shifting like mini rainbows. But what really struck me was how fragile they were. One wrong gust of wind or too much excitement from our laughter and—pop! Gone in an instant.
Now, let’s talk about why this matters beyond just fun playdates or childhood memories. Bubbles are used in all sorts of fields! For example, they play a role in firefighting by creating foam that smothers flames. That bubbling foam is effective because it cuts off oxygen from the fire while also cooling down the area – a nifty trick if you ask me.
Then there’s medicine! Scientists are experimenting with microbubbles made from lipids to help deliver drugs more precisely within our bodies. Imagine tiny bubbles carrying medicine straight to the cells that need it. It could change how we treat illnesses entirely!
And don’t get me started on bubble wraps! They make moving exciting (and less nerve-wracking). Ever heard that satisfying pop? That sound is synonymous with safety!
So yeah, while blowing bubbles might feel like sheer playtime bliss, there’s real science behind them that spills into various applications we encounter every day—from fighting fires to high-tech medicine. Makes you appreciate those colorful little spheres even more—don’t you think?