So, picture this: you’re at a party, right? Everyone’s mingling, and someone casually mentions hemoglobin. Cue the crickets, right? But hear me out! Hemoglobin is pretty much the VIP of your blood.
It’s like a lifesaver that carries oxygen around your body, making sure you don’t just collapse on the floor mid-conversation. Crazy important stuff, I’d say!
But what’s wild is that this little molecule is packed with secrets. Yeah, like how it twists and turns to do its job. Ever thought about how proteins are shaped? Well, it’s not just about what they do; it’s also about how they look.
So let’s chat about the structure of hemoglobin. It’s a fascinating journey through tiny parts that make a huge difference in our lives—without us even knowing it! You in?
“Exploring the Techniques Used to Determine the Structure of Hemoglobin in Biochemistry”
So, let’s talk about hemoglobin! You know, that amazing protein in your red blood cells that carries oxygen throughout your body. It’s kind of like a little delivery truck for oxygen. The structure of hemoglobin is crucial for its function, and figuring out what it looks like at the molecular level has been a big deal in biochemistry.
First off, the main techniques used to determine the structure of hemoglobin include X-ray crystallography, NMR spectroscopy, and cryoelectron microscopy. Each of these methods sheds light on how this protein is built—a bit like piecing together a puzzle.
X-ray crystallography is one of the most common techniques. Researchers first have to crystallize the hemoglobin. Sounds easy, right? Well, it’s tricky! Once they get good crystals, they hit them with X-rays. The way these rays bounce off the atoms gives scientists clues about the arrangement of those atoms in space. Think of it as shining a light on a hidden picture; you start seeing details you can’t catch with just your eyes.
Then there’s NMR spectroscopy. This one’s super cool because it happens in solution—like, you don’t have to make crystals! NMR uses strong magnetic fields to look at the nuclei of certain atoms—usually hydrogen or carbon. When placed in a magnetic field, those nuclei end up giving off signals that scientists can translate into structural information about the molecule. It’s pretty wild how you can gather so much info without solid forms!
Next up is cryoelectron microscopy, or cryo-EM for short. With this method, researchers freeze their samples really quickly so they preserve their natural state without forming ice crystals. They basically take pictures from different angles using electrons instead of light (because electrons have much shorter wavelengths). This helps them visualize large complexes like hemoglobin without needing crystallization!
But here’s where it gets personal: Imagine if scientists didn’t figure this stuff out! I remember hearing about a friend who suffered from sickle cell anemia—a condition caused by a tiny change in hemoglobin’s structure. Understanding what goes wrong at that molecular level has opened doors for potential therapies.
So yeah, determining hemoglobin’s structure isn’t just some abstract concept; it has real-world implications! The combination of these techniques allows researchers to create detailed models which help us understand how oxygen binds and gets released in our bodies effectively.
To wrap this up:
- X-ray crystallography: Great for detailed atomic arrangements.
- NMR spectroscopy: Works well in solution; no crystals needed!
- Cryoelectron microscopy: Preserves natural states and reveals large structures.
All these methods together have painted an incredible picture of hemoglobin’s architecture and function, helping us appreciate just how special our bodies are at getting oxygen to where it’s needed!
Exploring the Molecular Structure of Hemoglobin: How Its Design Supports Oxygen Transport in Biological Systems
Alright, let’s talk about hemoglobin! You know, that amazing protein in your red blood cells? It’s like the delivery truck for oxygen in your body. Seriously, without it, we wouldn’t last long.
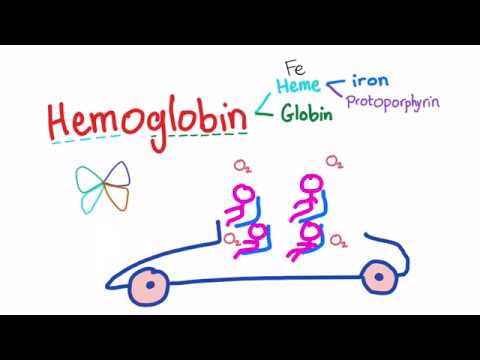
So, what’s the deal with its molecular structure? Well, hemoglobin is made up of four protein subunits. Think of each subunit as a piece of a puzzle. These subunits are called globins, and they come together to form a larger structure. Each globin has a heme group attached to it. And here’s the kicker: heme is what actually grabs onto oxygen!
- Heme Group: This little section contains iron at its center. Iron is super picky and loves to bond with oxygen. So when hemoglobin travels through our lungs, it picks up oxygen molecules because the iron in heme wants some new buddies.
- Quaternary Structure: Hemoglobin has this fancy setup called quaternary structure because it’s made of those four globin chains. This arrangement isn’t just for show; it’s crucial for how hemoglobin works. When one of those chains binds to oxygen, it actually changes shape slightly, making it easier for the others to bind more oxygen too!
- Cooperativity: The fancy term for this shape-change phenomenon is cooperativity. So basically, when one oxygen molecule hops on board, it’s like saying “Hey everyone! Come join us!” It makes it easier for more oxygen to attach.
You might be curious about why this matters so much. Well, look around you; every time you take a breath in, you’re providing your blood with fresh oxygen so your body can function well! Hemoglobin makes sure that as you move from high-oxygen areas (like your lungs) to lower-oxygen areas (like your muscles), it can release that oxygen right where it’s needed most.
A neat little detail? Hemoglobin can also bond with carbon dioxide—a waste product from our cells—helping carry it back to the lungs where we breathe it out. It’s like a two-for-one deal! Talk about multitasking.
The design of hemoglobin is truly remarkable—you could say it’s like nature’s perfect solution for oxygen transport in living systems. Just imagine if each cell had its own little delivery truck: chaos everywhere! Instead, we have hemoglobin working hard for us with elegance and efficiency.
This whole process highlights just how beautifully complex life is at a molecular level. Every breath you take isn’t just about staying alive; it’s also about this intricate dance happening inside you all the time!
Understanding the Molecular Structure of Hemoglobin: Key Features and Functions in Biochemistry
Hemoglobin is like the superstar of our blood. This protein, found in red blood cells, plays a key role in how we transport oxygen throughout the body. When you look at its molecular structure, you see something quite fascinating!
First off, hemoglobin is made up of four protein chains. You’ve got two alpha chains and two beta chains. Each of these chains wraps around a central iron-containing molecule called heme. And this heme is where the magic happens! It’s the part that actually binds to oxygen.
The cool thing about this structure is that it can change shape. When one heme binds to an oxygen molecule, it causes the whole hemoglobin protein to change slightly. This makes it easier for the next heme to grab its oxygen too! It’s kind of like a team high-fiving; once one gets going, the others follow along.
This unique property is known as cooperativity. It means that hemoglobin becomes more efficient at picking up and releasing oxygen depending on where it is in the body. For instance, in your lungs, there’s a lot of oxygen available, so hemoglobin happily fills up. But once it moves into tissues where oxygen is low (like your muscles after running), it releases that oxygen quickly. Pretty neat, right?
You might also be curious about how all this works biochemically. Hemoglobin doesn’t just bind to any gas floating around; it specifically carries oxygen (O2). But there’s more! Hemoglobin can also transport carbon dioxide (CO2) back to your lungs for exhalation, as well as protons which help maintain pH balance in your blood.
- The heme group: Each heme can bind one molecule of oxygen.
- The alpha and beta chains: They help stabilize each other and facilitate the changes needed for cooperativity.
- The iron atom: This central atom within each heme group is what makes binding of oxygen possible—iron loves to latch on!
- P53 mutation link: Some diseases arise from mutations in hemoglobin genes, affecting how efficiently it functions.
This whole setup means our bodies can adapt well to different activities. If you’re chilling on the couch or doing an intense workout, your body adjusts how much oxygen gets delivered exactly when and where it’s needed.
A little personal story: I once watched a documentary about divers who go deep underwater without tanks. They hold their breath for ridiculously long times due to something called myoglobin in their muscles—similar to hemoglobin but designed for muscle cells instead. The way these molecules function so effectively under pressure was mind-blowing!
This just goes to show you how amazing these tiny structures are! So next time you feel out of breath or after an intense jog, remember—your body’s incredible system of hemoglobin is working hard behind the scenes.
So, let’s talk about hemoglobin for a second. You know, that cool protein in your red blood cells that carries oxygen around your body? It’s kind of like the Uber driver for oxygen molecules, but way more complex and crucial.
I remember when I first learned about hemoglobin in school. We had this hands-on experiment where we dyed a piece of fabric with food coloring to visualize how oxygen binds to it. It was kind of messy, but watching those colors blend felt magical! That idea—how something so vital is hidden within us—really hit home.
Now, breaking down the molecular structure, hemoglobin’s made up of four subunits, each containing an iron atom. Picture this: each iron can snag one oxygen molecule. That means one hemoglobin can carry four oxygen molecules at a time! Isn’t that just insane? You’ve got these globular protein structures working tirelessly while you go about your day.
But wait, there’s more! Hemoglobin isn’t just like a sponge soaking up oxygen; it changes shape when it binds to these molecules. This property is called cooperative binding—in fancy terms—but really it’s just hemoglobin being social, adapting based on how much oxygen is around. If you’re exercising and need more oxygen? Hemoglobin responds by grabbing onto it even tighter!
And you know what’s even cooler? When hemoglobin releases those oxygen molecules back to your tissues—like little delivery robots—it actually tightens up again. This dance ensures you get enough of what you need when you need it most.
I guess what I’m trying to say here is that while we might not always think about what’s happening inside our bodies, there are these tiny miracles going on all the time. Hemoglobin might seem small on a molecular level, but its role in our survival is nothing short of heroic. Just imagine how many times today yours has ferried life-sustaining oxygen through your veins while you’ve been reading this!