You know that moment when you’re trying to identify a weird smell in your kitchen? Like, is that burnt toast or something else entirely? Well, IR spectroscopy is kinda like that, but for scientists!
Picture this: You shine infrared light on a sample and watch it dance. Sounds cool, right? This technique helps researchers figure out what’s really going on at the molecular level. It’s all about understanding how different molecules absorb light and what that says about them.
It’s not just for labs either. From pharmaceuticals to food science, IR spectroscopy gets around! It’s like the quiet hero of scientific research. So, whether you’re sniffing out substances or investigating complex compounds, this tech has got your back. Let’s see how it works and why it’s so darn useful!
Exploring the Role of IR Spectroscopy in Scientific Research: Applications and Insights
Alright, so let’s chat about IR spectroscopy, shall we? It’s one of those cool tools that scientists use to figure out what’s going on at the molecular level. Imagine it as a super-sophisticated way of using light to examine materials. Basically, it works by shining infrared light onto a sample and then studying how that light interacts with the molecules in the material.
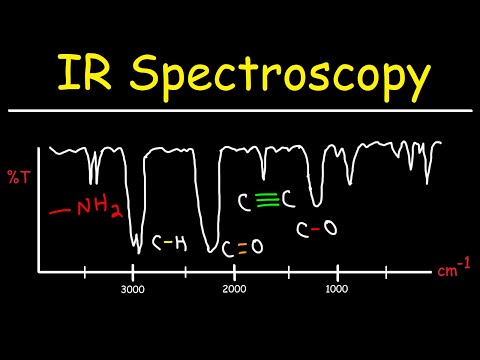
The thing is, different molecules absorb different wavelengths of infrared light. This absorption creates a sort of “fingerprint” for each substance. When you grab an IR spectrum—that’s just the output from the technique—you can see peaks that correspond to specific vibrations of chemical bonds within the molecules. It’s like listening closely to a song and picking out individual instruments!
Now, let’s look at some places where IR spectroscopy really shines in scientific research:
- Chemistry: In organic chemistry labs, researchers often use IR spectroscopy to identify functional groups in compounds. For example, if you synthesize a new molecule and want to check if you’ve got an alcohol or an acid on your hands, running it through IR will show unique signals for -OH or -COOH groups.
- Biochemistry: Here, it helps analyze biological samples like proteins and lipids. Think about studying enzyme structures—IR can provide insights into secondary structure elements like alpha-helices and beta-sheets. Ever had that moment when you’re waiting for your bread to rise? Well, scientists are doing something similar with proteins—they need time and conditions to change shape.
- Environmental Science: Scientists monitor pollutants in air or water using IR spectroscopy, making it easier to detect harmful substances. Let’s say they want to find out if there’s too much methane floating around; they can shine IR light on the sample and get results pretty fast!
- Material Science: Here’s where things get super interesting! Researchers can assess polymers or nanomaterials by evaluating their molecular composition via IR spectra. Imagine designing stronger materials for better smartphones or lighter airplane parts—this tech is key in characterizing them.
But wait, there’s more! One incredible aspect of IR spectroscopy is its ability to monitor changes over time. Picture this scenario: you’re baking cookies and you want to know when they’re just right without sticking your nose in (though how could anyone resist?). Scientists can do something similar by tracking real-time reactions using IR spectra.
You might wonder about limitations; well, like any tool, it has a few quirks. Sometimes overlapping signals make it tricky to interpret data accurately. Or if a sample is too complex? Yeah, good luck untangling that mess! It requires some expertise—kind of like being able to read between the lines in a mystery novel.
Overall, whether it’s determining chemical structures or keeping our environment healthy, IR spectroscopy plays a crucial role across various fields of scientific research. So next time someone mentions how they analyzed something using this nifty technique, you’ll totally know what they mean! Cool stuff happening under that mysterious spectrometer hood!
Applications of IR Spectroscopy in Forensic Science: Enhancing Crime Scene Analysis and Evidence Identification
Forensic science, like a captivating mystery novel, often hinges on tiny clues that can shed light on what really happened. One such tool in the forensic toolbox is infrared (IR) spectroscopy, a technique that’s super useful when it comes to analyzing different substances found at crime scenes.
So, what exactly is IR spectroscopy? Well, it’s a method that measures how molecules absorb infrared light. This absorption happens at specific wavelengths based on the chemical bonds within the molecules. Think of it like each substance has its own unique fingerprint. When you shine infrared light on a sample, the way it absorbs that light tells scientists a lot about its identity.
Now, let’s look at some key applications of IR spectroscopy in forensic science:
- Identifying Drugs: One of the most common uses is in identifying illicit drugs. Forensic experts can analyze powdery substances and determine what they are quickly and accurately.
- Aids in Analyzing Paint: If there’s paint residue from a vehicle at a crash site or crime scene, IR spectroscopy can help identify the type and even brand of paint. This can link suspects to crimes.
- Explosive Residue Detection: After an explosion, scientists can use this technique to analyze debris for traces of explosive materials. It helps pinpoint what kind of explosive was used.
- Analyzing Fibers and Textiles: Fibers from clothing or carpets can provide clues as to who was present during a crime. IR spectroscopy helps differentiate between different types of fibers based on their chemical structure.
I once read about this fascinating case where detectives were trying to solve a burglary. They found tiny blue fibers clinging to some broken glass at the scene. Using IR spectroscopy, they matched those fibers to a rare fabric from a designer suit—leading them right to their suspect! It’s pretty mind-blowing how something so small could play such a huge role in solving cases.
But it doesn’t stop there! IR spectroscopy can also be applied in other areas like determining the composition of unknown substances or even analyzing biological materials—like sweat or saliva found at crime scenes—which might hold DNA evidence.
So yeah, while it might seem like just another fancy lab tool, IR spectroscopy is seriously influential in forensic science! By helping identify materials and link them to potential suspects or events, it enhances crime scene analysis and plays an essential part in uncovering the truth behind criminal activities.
Exploring the Nature of IR Spectroscopy: Qualitative vs. Quantitative Analysis in Scientific Research
So, IR spectroscopy! It’s that cool technique where scientists use infrared light to understand what molecules are made of. If you’ve ever wondered how chemists figure out the structure of a compound or detect a specific substance in a mixture, this is one way they do it. Let’s dig into it.
What is IR Spectroscopy?
IR spectroscopy works by sending infrared light through a sample. Molecules absorb some of this light based on their unique vibrations, like how you might sway your body when you hear music. The resulting spectrum is basically a fingerprint for that molecule, showing which wavelengths were absorbed.
Qualitative Analysis
When we talk about **qualitative analysis**, we’re looking to identify the components present in the sample. Imagine you’re at an ice cream shop with all those flavors. You can tell chocolate from vanilla just by tasting them, right? That’s kind of what qualitative IR spectroscopy does for molecules.
When you get an IR spectrum, you’ll see peaks that correspond to various molecular vibrations. For example:
- The O-H bond from alcohols shows up as a strong, broad peak around 3200-3600 cm-1.
- C=O stretches appear typically around 1700 cm-1.
From these peaks, scientists can infer what compounds are in their sample. It’s like piecing together clues from a mystery novel but with chemical bonds instead!
Quantitative Analysis
Now let’s switch gears to **quantitative analysis**. This part is all about measuring how much of something is there—like counting the scoops of ice cream you bought. In IR spectroscopy, we use the intensity of those peaks I mentioned earlier to figure out concentrations.
For instance, if you know that a certain peak’s intensity relates directly to the concentration of a compound in your sample (thanks to something called Beer-Lambert Law), you can measure just how much there is! Like if you’re testing blood glucose levels; more glucose means stronger absorption at specific wavelengths.
The Balance Between Qualitative and Quantitative
Both these types are used together quite often. Let’s say you’re analyzing environmental samples for pollutants. First, you’d identify which harmful chemicals are present (qualitative). Then you’d measure how much of each pollutant exists (quantitative). This dual approach gives researchers valuable insights into environmental safety and health risks.
Applications in Real Life
Think about perfume makers who want exactly the right scent or doctors needing to analyze bodily fluids for diagnoses—IR spectroscopy plays a huge role everywhere from labs to factories.
In academia or industry settings, researchers use it for everything from studying complex biological materials to ensuring quality control in pharmaceuticals and food products.
So basically, whether you’re trying to sniff out what’s inside a mysterious liquid or measure exact amounts for medical tests, IR spectroscopy has got your back! With its ability to analyze both qualitatively and quantitatively, it bridges gaps in scientific research like few other methods can.
So, you know when you’re trying to figure out what something smells like? Like when you walk into a bakery and get that warm, buttery scent of fresh croissants wafting through the air? It’s kind of like your brain is pulling together bits of information to recognize the sweet goodness. That’s similar to what IR spectroscopy does but with molecules instead of scents.
IR (or infrared) spectroscopy is this cool technique scientists use to identify different substances based on how they absorb infrared light. You can think of it as shining a flashlight on a mystery object and seeing which colors get absorbed or reflected. Each molecule has its unique fingerprint—like our own, but for chemicals. When those molecules absorb infrared light, they start to vibrate in specific ways. By measuring these vibrations, scientists can figure out what the molecule is and even how it’s structured.
I remember one time in a lab, we were working on analyzing some unknown samples for a project. The atmosphere was filled with excitement—everyone was curious about what we were going to find out. After running the IR spectroscopy analysis, we uncovered the presence of an unexpected component in our sample! It felt like uncovering buried treasure; everybody was buzzing with ideas on how this new bit could change everything we thought we knew about it.
In research, IR spectroscopy shines brightly in diverse fields: chemistry, environmental science, even medicine! It helps identify pollutants in air or water—a huge deal for keeping our planet healthy—or it can reveal details about unknown compounds that might play essential roles in new drugs. And let me tell you, having reliable methods for identifying materials quickly can save researchers so much time.
But here’s what I find truly interesting: while the tech behind IR spectroscopy is advanced, it doesn’t feel intimidating once you get into it. At its core, it’s just about understanding how light interacts with matter—something so fundamental that even kids can grasp if explained simply enough. That connection makes science feel accessible and relatable.
So yeah, IR spectroscopy isn’t just some lab technique gathering dust on a shelf. It’s an active part of scientific discovery that opens doors and sparks questions in all sorts of fields. Just like those delightful bakery smells lead us to delicious treats; understanding molecular identities leads researchers down paths filled with exciting possibilities!