You know that moment when you’re trying to figure out a puzzle, and the last piece just won’t fit? Well, that’s kinda how chemists feel about atoms sometimes. It’s like they’ve got this wild party going on, and every guest needs to be in the right spot for it to work.
Now, let’s talk about Gilbert Lewis. Picture a young guy in a lab coat scribbling away, trying to make sense of this atomic chaos. He came up with this atom model that changed the game. Seriously!
The Lewis atom model is not just some dusty old theory; it’s like the blueprint for so much of modern chemistry today. It helps us understand everything from why salt tastes salty to how our bodies process vitamins.
So buckle up! We’re diving into how this simple concept has shaped our world in ways you probably never thought about.
Understanding Lewis Structures: Their Significance in Chemistry Explained
Understanding Lewis Structures is like getting a backstage pass to the world of molecules. So, grab a snack and let’s break it down!
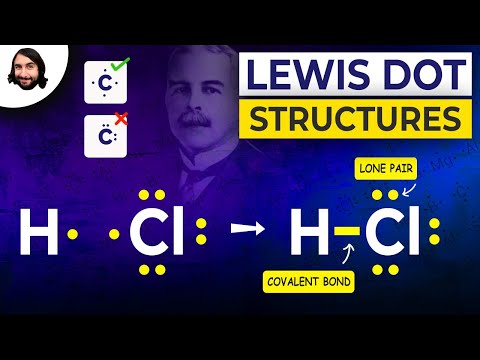
First off, what are Lewis structures? They’re pretty much diagrams that show how atoms bond in a molecule. Named after Gilbert Lewis, who introduced them way back in 1916, these structures help visualize the arrangement of electrons around atoms. You know those dots you used to draw in art class? Well, Lewis structures use dots for valence electrons, which are the outermost electrons that play a key role in bonding.
Now, why do we care about these little guys? Understanding bonding is crucial because it explains how substances interact with one another. For example, take water (H₂O). In its Lewis structure, you can see how oxygen shares electrons with hydrogen atoms to create those cool water molecules we all love so much.
But hold on— there’s more! Not only do these structures help us see bonds, but they also show us the shapes of molecules. Each atom’s position affects everything—from boiling points to reactivity. When you sketch out a Lewis structure for carbon dioxide (CO₂), you’ll notice it has a linear shape due to how the electrons bond and repel each other.
One nifty thing is recognizing whether bonds are single or multiple. When an atom shares more than one pair of electrons with another atom? Yep! That’s where double or triple bonds come into play. For example, in ethene (C₂H₄), there’s a double bond between carbon atoms that changes how the entire molecule behaves.
The rules for drawing these structures are fairly straightforward but require practice. Here’s what you typically do:
- Count the valence electrons: Start by adding up all the outer shell electrons from each atom.
- Create bonds: Connect atoms with lines representing shared pairs of electrons.
- Add lone pairs: Place any leftover electrons around the atoms until each one has an octet (or duet for hydrogen).
- Check your structure: Make sure everything follows the rules and makes sense chemically.
So here’s an emotional snapshot—imagine sitting in chemistry class feeling totally lost while your teacher scribbles complex formulas on the board. But then they introduce you to Lewis structures! It’s like someone flipped on a light switch! Suddenly you can visualize electron interactions and grasp why certain chemicals react differently.
Finally, understanding these structures is like having a toolbox for tackling chemical reactions and predicting outcomes. Whether you’re studying basic compounds or delving into complex organic chemistry, Lewis structures will be your friend. Just remember: they give insights not only into individual molecules but also into larger concepts like resonance and formal charges.
In short, mastering Lewis structures opens up a whole new layer of understanding in chemistry—it helps connect dots literally and figuratively! So next time someone mentions them at a party (because wouldn’t that be wild?), you’ll know just what they’re talking about!
The Significance of the Lewis Model of the Electron in Advancing Chemistry and Scientific Understanding
The Lewis model of the electron is a big deal in chemistry. It helps us understand how atoms behave, especially when they’re forming bonds. But what exactly does it mean? Let’s break it down.
First off, the Lewis structure, which you might know as Lewis dot diagrams, shows how electrons are arranged around an atom. Basically, you draw dots around the chemical symbol to represent electrons. More dots mean more electrons. Pretty straightforward, right?
Now, the real significance comes into play when these dots help us understand covalent bonding. You see, atoms like to bond together to achieve a full outer shell of electrons—usually eight of them (yep, that’s called the octet rule). For example, when two hydrogen atoms (which each have one electron) come together with an oxygen atom (which has six), they form water (H₂O). In this case, sharing electrons helps everyone feel satisfied.
And it doesn’t stop there! The Lewis model is super useful for predicting how different elements will interact with one another. Like a chemistry detective! Just by looking at their valence electrons—the ones in the outermost shell—you can guess their behavior. For instance:
- Elements with similar numbers of valence electrons often behave similarly.
- The number of bonds an atom can form usually matches its available valence electrons.
This idea also opens the door to discussing things like ionic bonding. When an electron is transferred from one atom to another—like sodium giving an electron to chlorine—you create charged ions that attract each other. This concept was groundbreaking because it provided a clear visual way to represent these interactions.
So why should we care? Well, understanding these interactions laid down some serious groundwork for modern chemistry and materials science. Think about it: everything from antibiotics to solar panels involves these fundamental principles. The Lewis model made it easier for scientists and students alike to grasp complex reactions and even predict new chemical compounds.
Plus, there are tons of applications! In biochemistry or drug design, knowing how molecules will interact based on their Lewis structures can lead to smarter decisions when creating medications or other compounds.
In essence, without the Lewis model of the electron and its straightforward approach to visualizing atomic behavior and bonding strategies, our journey through chemistry would look very different today. It serves as both a teaching tool and a catalyst for advancements in scientific understanding! So next time you’re doodling some dots around atomic symbols in class or at home, remember that you’re tapping into something pretty big in the world of science!
Understanding Lewis Theory in Chemistry: Key Concepts and Applications in Chemical Bonding
Understanding Lewis Theory in Chemistry can really open your eyes to how atoms bond and interact with each other. So, let’s break it down into bite-sized pieces, shall we?
The guy behind this theory, Gilbert N. Lewis, introduced an idea back in the early 20th century that fundamentally changed chemistry. He proposed a way to visualize how atoms bond by looking at their electrons. Instead of diving into too much jargon, we can say his model uses symbols to represent atoms and their outermost electrons.
Now, why does the outermost layer of electrons matter? Well, these are called valence electrons, and they play a crucial role in chemical bonding. Atoms want to have a full outer shell of electrons, like the coveted eight. This is known as the octet rule. When atoms share or transfer these valence electrons, that’s where bonding happens.
There are two major types of bonds explained by Lewis’ theory:
Imagine you’ve got two friends who decide to share their pizza slices (that’s like a covalent bond). Or think about a friend giving away their last slice because they know the other friend is starving (that’s more like an ionic bond).
In drawing these connections out, Lewis created something called Lewis structures. These diagrams show how atoms are connected and provide a visual representation of electron sharing or transfer. You draw dots for valence electrons around chemical symbols. Lines between them represent shared pairs.
Let’s say we’re looking at water (H₂O). In its Lewis structure, you’d see the oxygen atom in the center—kind of like the host of a party—with two hydrogen atoms each giving up one electron to form shared pairs with oxygen. Pretty neat!
Now onto some important applications: Lewis Theory isn’t just for doodling in your notebook; it’s super useful! It helps predict things like
Also, this theory lays down the groundwork for understanding more complex laws in chemistry including resonance and hybridization later on in your studies.
So to sum it all up: Gilbert N. Lewis gave us tools to visualize how chemicals connect by focusing on those fabulous valence electrons. From drawing simple structures to predicting reactions, his ideas continue making waves in modern chemistry today—and that’s pretty cool!
Alright, so let’s chat about the Lewis Atom Model. You may have heard of it before, maybe even in a classroom setting, but it’s actually super interesting when you think about how it laid the groundwork for so much in modern chemistry.
First off, you know that moment in school when everything starts to click? For me, it was when we got to the part about bonding and molecules. The Lewis model really shines here. It’s all about how atoms bond by sharing or transferring electrons, which are these tiny particles buzzing around the nucleus of an atom. Think of them as tiny little dancers forming pairs! In Lewis’s world, these pairs are what create the bonds between atoms, paving the way for molecules—and ultimately everything we see around us.
Back in 1916 when Gilbert Lewis first introduced this model, he was trying to simplify things. He wanted a clear picture that would help explain why elements behave the way they do. And honestly? He nailed it! The use of dots to represent electrons surrounding symbols of elements makes it easier to visualize how atoms connect. Plus, it gives us insight into why certain atoms tend to combine more readily than others. Like oxygen and hydrogen forming water—it’s like a match made in heaven!
But here’s where my mind really goes wild: think about all the advances we’ve seen since then! Thanks to Lewis’s foundational ideas, chemists can explore complex molecules and even develop new materials. It feels pretty amazing that something so simple has had such a huge impact on science as we know it today.
And there’s something kind of poetic about that too, right? A simple idea leading to complex discoveries and innovations we couldn’t have imagined without those early models. It reminds me of how even small actions can create waves—like tossing a stone into a pond and watching the ripples spread.
Ultimately, while modern chemistry has evolved with advanced theories and tech tools, the charm of Lewis’s Atom Model still lingers on in our labs and classrooms today. It serves as a reminder that simplicity can be incredibly powerful—a bit like life itself! So next time you’re mixing things up in chemistry class or just exploring how things around you work at a molecular level, give a little nod to Gilbert Lewis and his eye-opening model because without it… well things might look pretty different!