So, picture this: you’re in a crowded room, and you spot one person gravitating towards another. They seem to vibe, right? That’s kind of how atoms work, too!
Atoms don’t just float around all lonely. They prefer to team up, forming bonds like friendships. Some are tight-knit, while others are more relaxed.
Let’s get into the two main types of bonds that make chemistry tick: covalent and ionic bonds. These guys are like the dynamic duo of the atomic world.
Trust me; once you grasp how these bonds come together—or clash—you’ll see chemistry isn’t just about boring equations. It’s like a relationship drama but with atoms instead of people! So grab your popcorn; we’re about to dig in!
Exploring the Three Main Types of Chemical Bonds in Science
So, let’s talk about chemical bonds. They’re super important in the world of chemistry and can be pretty interesting! What you need to know is that there are mainly three types of chemical bonds: covalent bonds, ionic bonds, and metallic bonds. Each type has its own way of forming and they play a big role in how atoms interact.
Covalent Bonds are like a friendly handshake between two atoms. When two non-metal atoms come together, they share electrons to fill their outer shells. Imagine you and a friend want to hold hands while walking; that’s kind of what happens with these atoms! For example, in a water molecule (H₂O), each hydrogen atom shares one electron with the oxygen atom. So here you get two hydrogens holding on tight to one oxygen!
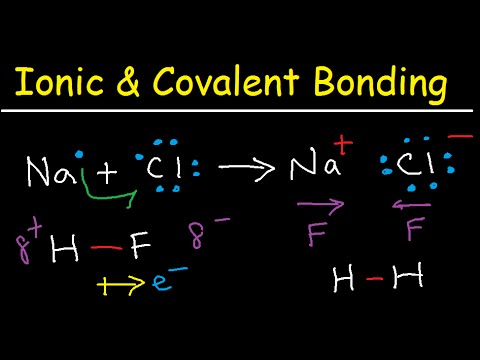
Now, on to Ionic Bonds. This type is like a game of tug-of-war between two different elements—typically a metal and a non-metal. One atom gives up one or more electrons (let’s say it’s the metal), so it ends up with a positive charge. The other atom takes those electrons (that’s usually the non-metal) and ends up negatively charged. They’re now like magnets attracting each other! A classic example is sodium chloride, or table salt, where sodium gives away an electron to chlorine.
Then there’s Metallic Bonds. If covalent bonds are hand-holding and ionic bonds are tug-of-war, then metallic bonds feel more like a big group hug! In this case, metal atoms share their electrons freely among many other metal atoms—kind of like passing around snacks at a party. This “sea of electrons” makes metals good conductors of electricity and gives them their shiny appearance.
So yeah, these three types really shape how substances behave and interact with each other:
- Covalent Bonds: Sharing electrons among non-metals.
- Ionic Bonds: Transfering electrons between metals and non-metals.
- Metallic Bonds: Sharing electrons among many metal atoms.
Next time you toss some salt on your fries or pour yourself a glass of water, just remember; there’s quite the chemistry happening behind the scenes! It’s all about those little connections that keep everything together in this wild world we live in.
Exploring Ionic and Covalent Compounds: 5 Key Examples in Chemistry
Sure, let’s get into the nitty-gritty of ionic and covalent compounds. Chemistry can seem a bit intimidating sometimes, but it’s honestly just a matter of understanding how atoms hang out together. Think about it as atoms having a party—some prefer sharing snacks while others go for trade-offs. So, let’s break this down!
Ionic Compounds are formed when atoms transfer electrons. Picture two friends: one has extra cookies and the other is hungry. The cookie-rich friend gives away some treats, while the other happily takes them. This transfer creates charged particles or ions—one becomes positively charged (cation) and the other negatively charged (anion). These opposite charges attract each other, binding them like best pals.
Examples of Ionic Compounds:
- Sodium Chloride (NaCl): Common table salt! Sodium donates an electron to chlorine, creating Na⁺ and Cl⁻ ions.
- Calcium Fluoride (CaF₂): Here, calcium shares two electrons with two fluorine atoms—you get one positive Ca²⁺ ion and two negative F⁻ ions.
- Magnesium Oxide (MgO): Magnesium donates its two electrons to oxygen, forming Mg²⁺ and O²⁻ ions.
Now let’s chat about Covalent Compounds. Unlike ionic bonding, this is all about sharing. Imagine two friends splitting a pizza evenly. They both take slices from the same pie—this is how covalent bonds work; atoms share pairs of electrons to achieve stability.
Examples of Covalent Compounds:
- Water (H₂O): Two hydrogen atoms share their electrons with one oxygen atom—voilà! You have water, essential for life!
- Carbon Dioxide (CO₂): Here, carbon shares its four electrons with two oxygen atoms; it makes that classic gas we exhale!
- Methane (CH₄): In this case, carbon shares its four electrons equally with four hydrogen atoms for a happy molecule.
It’s kind of cool how those bonds work together in our daily lives! Like when you sprinkle salt on your food or take a sip of water; you’re experiencing chemistry at play in delicious action.
So remember: ionic compounds are about giving away and attracting charges while covalent compounds are all about sharing nice and evenly. It’s all about building those connections—whether they’re tight ones like in sodium chloride or cozy ones like in water. Chemistry isn’t just formulas; it’s really all around us!
Understanding Covalent and Ionic Bonds: Key Questions in the Foundations of Chemistry
Understanding Covalent and Ionic Bonds is like peeling an onion—you’ve got layers to uncover, and it can make you shed a few tears if you don’t get it right. So, let’s break this down, nice and simple.
Covalent bonds are your friendly neighborhood connections. They happen when two atoms decide to share their electrons. Imagine you and a friend want to play a game, but there’s just one controller. Instead of fighting over it, you both take turns using it together. In chemistry terms, when atoms share electrons like that, they form a covalent bond.
For example, look at water (H₂O). Each hydrogen atom shares its electron with the oxygen atom so that everyone is happy and stable—just like a cozy family game night!
On the flip side, we’ve got ionic bonds. These are more like a tug-of-war match where one side wins big time. In ionic bonding, one atom gives up an electron to another atom. This creates ions; one becomes positively charged and the other negatively charged—kinda like magnets that attract each other.
Take sodium chloride (table salt) as an example. Sodium (Na) loses an electron and turns into Na⁺ (a positive ion), while chlorine (Cl) gains that electron making Cl⁻ (a negative ion). They stick together because opposites attract! It’s all about balance in their little world.
Now, let’s highlight some key differences:
- Sharing vs Giving: In covalent bonding, atoms share electrons. In ionic bonding, one atom gives away an electron completely.
- State of Matter: Covalent compounds often exist as gases or liquids at room temperature; ionic compounds are typically solid crystals.
- Conductivity: Ionic compounds conduct electricity when dissolved in water because those ions can move freely. Covalent compounds usually don’t conduct electricity.
So why do we care? Well, these bonds are fundamental in chemistry; they determine how molecules behave and interact with each other. Understanding them helps us grasp everything from how our bodies function to how different materials react around us.
You know? Once I tried to make lemonade without knowing about these bonds—the sugar didn’t dissolve properly! It was a bummer because I didn’t understand why things weren’t mixing well. Turns out ionic compounds like table salt dissolve easily in water while some covalent compounds don’t mix as well.
In summary: Covalent bonds are all about sharing—two atoms working together for mutual benefit; while ionic bonds show us what happens when someone gives up something for the greater good of bonding with another atom! Both types of bonds create unique behaviors for elements that ultimately shape the world around us. Isn’t chemistry cool?
So, let’s chat about covalent and ionic bonds. You know, these little things are like the glue holding everything together in chemistry. Imagine walking through a park, and you see kids playing on swings. That connection between the swing and the chains is a lot like how atoms bond with each other.
Covalent bonds happen when atoms share electrons. It’s like two best friends sharing their favorite toys. They’re both getting something out of it, and it creates a strong bond that keeps them close. For example, take water—H2O! Each hydrogen atom shares its electrons with an oxygen atom, and boom! You’ve got water. Without those covalent bonds, we’d be missing out on one of life’s essentials.
Now, ionic bonds are a bit different; they remind me of an intense game of tug-of-war. Here’s how it works: one atom basically steals an electron from another. This creates positively charged ions and negatively charged ions that attract each other like magnets. Sodium chloride (that’s table salt) is a classic example. Sodium gives up an electron to chlorine, making them stick together in this really cool way.
Thinking back to my school days, I remember sitting in chemistry class trying to grasp these concepts while staring at diagrams in our textbook. It felt overwhelming at first—like learning a new language! But then we did this experiment where we actually made salt crystals from sodium and chlorine. Watching those tiny crystals form felt magical; I finally understood why these bonds matter!
Ultimately, these two types of bonds are foundational for everything around us—plants growing, water flowing, you name it! And while they might seem straightforward on the surface (no pun intended), they create such complex structures that breathe life into our universe.
So the next time you’re drinking some water or seasoning your food with salt, just know there’s some serious bonding going on at the atomic level! Seriously cool stuff when you think about it!