So, picture this: you’re at a party, and someone starts talking about covalent bonds. Like, how often does that happen, right? You might find yourself nodding along, but inside you’re thinking, “What on Earth are they talking about?”
Well, fear not! Covalent bonds are kinda like that secret sauce in a recipe. They hold things together in ways you might not even realize. It’s not just about atoms; it’s about how they play nice and share their toys—like electrons.
And guess what? This whole sharing thing is way more important than it sounds. It’s basically the backbone of all the cool stuff around us! From water to air to your favorite snack—yeah, those tasty chips probably owe their flavor to some nifty covalent bonds.
Come with me on this journey into the world of covalent bonds. We’ll break it down together, no PhD required!
Understanding Covalent Bonds: A Comprehensive Guide to Chemistry in Science
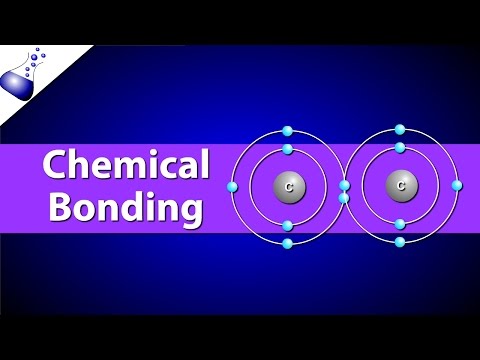
Covalent bonds are like the ultimate friendship in chemistry. You know how friends sometimes share their last slice of pizza? Well, atoms do something similar with their electrons! Instead of just hanging out alone, they share electrons to fill up their outer shells and feel stable. It’s this sharing that creates a strong bond between them.
When we talk about covalent bonds, we often think of molecules like water (H₂O) or carbon dioxide (CO₂). In water, each hydrogen atom shares one electron with the oxygen atom. This sharing makes the water molecule super special and essential for life. Without it, well, no swimming pools!
Now, there are different kinds of covalent bonds, which makes things even more interesting. Take a look:
- Single Bonds: This is when two atoms share one pair of electrons. Like in H₂ (hydrogen gas), where two hydrogen atoms bond by sharing one pair.
- Double Bonds: Here, two pairs of electrons are shared between atoms. For instance, in O₂ (oxygen gas), each oxygen atom shares two pairs of electrons.
- Triple Bonds: Now we’re getting serious! Three pairs of electrons are shared here, like in N₂ (nitrogen gas). This is a strong bond that keeps nitrogen molecules together.
The strength of these covalent bonds can vary too! Single bonds are usually the weakest, while triple bonds are the strongest. It’s like comparing a light hug to a bear hug—one is gentle and easy to break, while the other holds on tight!
You might be wondering what happens when these covalent bonds form shapes or structures? Well, that’s where things get really cool. The arrangement of bonded atoms affects the properties of substances dramatically. For example, carbon can form four covalent bonds and create amazing structures like diamonds—which are super hard—or graphite—which is soft and used in pencils.
And here’s something you might not think about: not all electron sharing is equal! Sometimes one atom pulls harder on the shared electron than another atom does because it has a stronger pull (we call this electronegativity). This unequal sharing creates what we call polar covalent bonds, like in water again! The oxygen end gets slightly negative while the hydrogen ends become slightly positive.
You see how complex and beautiful it can be? It’s almost as if each type of bond tells its own story about how elements come together and behave differently in our world. So next time you take a sip of water or draw with a pencil, remember that at the atomic level there’s a lot more going on than meets the eye!
Understanding Covalent Bonds: Do They Share or Donate Electrons in Molecular Chemistry?
So, let’s chat about covalent bonds. You might have heard about them in chemistry class or while trying to piece together how molecules actually work. But what really happens when atoms get together? Do they share or donate electrons? Well, buckle up because we’re diving into this!
Covalent bonds are all about sharing. Imagine two friends who decide to share their snacks instead of just keeping them to themselves. In the world of atoms, when two non-metal atoms come close enough, they start to “share” electrons. This happens because each atom wants a full outer shell of electrons to feel stable—like having the ultimate party vibe!
Let’s break it down a bit. You see, every atom has a certain number of electrons. These are arranged in layers around the nucleus—kind of like floors in a building. The outer layer is super important because it determines how atoms interact with each other. Atoms strive for this magical number called the octet rule, where they want eight electrons in their outer shell (there are some exceptions, but we’ll keep it simple for now).
Now, when two atoms decide to bond covalently:
- They each contribute one or more of their own electrons.
- The shared pair(s) fill up both their needs.
This is what makes them happy little molecules! A classic example is water (H₂O). Here’s how that works: An oxygen atom needs two more electrons to fill its shell while each hydrogen atom has one extra electron ready to share. So they all come together and share! Voilà! You have water.
But does that mean they’re giving away their electrons? Nope—not really! They’re more like saying, “Hey, let’s hang out and share for a bit.” Both atoms benefit from this arrangement without anyone feeling left out.
Now you might wonder: What happens if one atom wants those shared electrons more than the other? That brings us to something called polarity. In cases like water again, oxygen is way better at snagging those shared electrons because it has a stronger pull due to its higher electronegativity—fancy word alert! This means oxygen becomes partially negative while hydrogens become slightly positive. It’s like the friendship between them is unequal but still working out!
And don’t forget about multiple covalent bonds! Some atoms get extra close and end up sharing more than one pair of electrons. For instance, in carbon dioxide (CO₂), each oxygen shares two pairs with carbon—which gives us double bonds!
So yeah, covalent bonding is all about cooperation between atoms through sharing rather than giving away or donating electrons entirely. It creates strong connections that allow molecules to form everything around us—from the air we breathe to our very own cells.
In short:
- Covalent bonds involve sharing not donating.
- Atoms want full outer shells; sharing gets them there.
- Polarity can affect how these relationships work.
- Molecules can use single or multiple pairs of shared electrons.
At the end of the day, understanding this concept opens up so many doors in chemistry and helps you appreciate just how interconnected everything really is—even at an atomic level! So next time you think of molecules bonding together, picture those little friends sharing snacks instead of giving them away entirely—it makes everything feel way more relatable!
Understanding Covalent Bonds: A Comprehensive Guide for IB Biology Students
Covalent bonds are like the strong friendship bonds between atoms. They happen when two atoms share electrons, which helps them fill their outer shells and feel more stable. So, you can think of it as two friends holding hands so they don’t get lost in a crowd.
Now, let’s break it down a bit more. When you look at atoms, they typically have protons, neutrons, and electrons. The electrons are the party animals that roam around the nucleus. Atoms really like to have a full outer shell of electrons, kind of like filling up your plate at a buffet! However, not all atoms have enough electrons by themselves to fill up that plate. That’s where covalent bonds come into play.
Covalent bonds can be classified into two main types: single bonds and multiple bonds. A single bond is when two atoms share one pair of electrons. Examples include hydrogen (H₂) and methane (CH₄). In these cases, it’s just one little dance move: one pair of hands holding each other tight.
Then there are double and triple covalent bonds. Double bonds involve sharing two pairs of electrons – you could imagine it as holding hands while hugging! An example would be oxygen gas (O₂). Triple bonds? Well, that’s three pairs being shared – like a group hug among friends! An example here is nitrogen gas (N₂).
Now, covalent bonding isn’t just about sharing; it’s also about how much each atom wants those shared electrons. This “want” is known as electronegativity. Some atoms are like super needy friends who want everything for themselves (*cough* fluorine *cough*), while others are more chill about sharing (*ahem* carbon). This neediness changes how strongly they hold onto the shared electrons.
In some cases, the sharing isn’t equal at all! When an atom with higher electronegativity pulls those shared electrons closer to itself, you get something called a polar covalent bond—like if one friend always tries to take the bigger slice of cake… Not cool! Water (H₂O) is a classic example here; oxygen hogs some electron love from hydrogen.
Why should you care about understanding these things? Well, knowing how covalent bonds work helps explain many biological processes happening in your body every day– from how proteins fold to how DNA holds genetic information together. It’s pretty neat stuff!
And lastly—that warm fuzzy feeling around these processes? It comes down to chemistry connecting our world in ways we often don’t see but totally rely on every day. Understanding covalent bonds gives you insight into life itself—from tiny cells buzzing with activity to massive ecosystems in balance.
So there you go! Covalent bonds help form the very basis for complex molecules that make life tick—pretty profound when you think about it!
Covalent bonds are one of those topics that, at first glance, can seem a bit like a puzzle. You know? It’s like when you were a kid trying to figure out how to connect the dots in a drawing. Once you get it, though, it becomes this beautiful picture that makes sense.
So, picture this: two atoms hanging out together. They’re not just chilling; they’re actually sharing something super valuable—electrons! That’s the essence of covalent bonds. Instead of ripping away electrons from each other (like in ionic bonds), they decide to share them. It’s all about teamwork—literally!
I remember my high school chemistry teacher explaining this concept with such enthusiasm. She had this way of making everyone feel excited about science; it was infectious! One time, she demonstrated how water molecules form by creating a mini-experiment with colored water and some balloons. The way those balloons hugged each other while sharing their water was just magical. It clicked for me at that moment: sharing is caring, even for atoms.
You see, covalent bonds are crucial for forming molecules we encounter every day—like H2O (that’s water!) or CO2 (carbon dioxide). They create everything from the air we breathe to the food we eat. It’s wild to think that our existence hinges on these tiny little connections.
Now let’s talk about types of covalent bonds. There are single, double, and even triple bonds! Think of them as friendship levels: you share one electron with a single bond, two with a double bond, and three with a triple bond. Pretty neat comparison, huh? Each adds strength and stability to molecules in different ways.
So why should you care about covalent bonds? Well, understanding them opens up a world where chemistry meets life itself. When you grasp how these connections work, it’s like having an insider’s view into nature’s blueprint.
And let’s be honest—it’s pretty cool when you realize every breath you take is part of this grand molecular dance happening all around us. You follow me? Covalent bonds might sound complex at first but once you break them down (no pun intended!), they reveal the amazing connections that make up everything!