You know that moment when you see a rainbow and can’t help but stare? It’s like nature’s own art show, right? Well, what if I told you that there’s a colorful world hiding in the elements around us? Seriously!
Picture this: each element on the periodic table has its own unique color signature. Yep, like a fingerprint but way cooler. Scientists can actually use these colors to figure out what stars are made of or even detect metals in a sample. How wild is that?
So, let’s chat about elemental spectra. It sounds fancy, but it’s really just about how light interacts with different elements. Trust me, it’s not as complicated as it sounds! Get ready to explore some seriously vibrant science!
Exploring the Science of Color: A Comprehensive PDF Guide to Chromatics and Perception
The world of color is truly mesmerizing, isn’t it? I mean, have you ever stopped to think about how you perceive the colors around you? It’s not just about looking at a rainbow or enjoying a sunset; there’s some serious science behind it. So let’s unravel this colorful mystery together!
Chromatics is the study of colors and how we see them. It combines physics, biology, and even a dash of psychology to explain what happens when light hits objects and then travels to our eyes. You know, light is made up of different wavelengths. Each wavelength corresponds to a different color on the visible spectrum. If you’ve ever used a prism to split light into colors, that’s chromatics at work.
Now, when light hits an object, some wavelengths are absorbed while others are reflected. This reflection gives objects their color! For instance, an orange typically reflects wavelengths that correspond with orange light and absorbs others. Fun fact: that’s why we see it as orange!
So how does this process play out in our brain? Well, it all starts in your eyes. Inside your retina are special cells called cones that are sensitive to different wavelengths of light—red, green, and blue. These cones send signals to your brain when they detect specific light waves.
Your brain takes this information and processes it so quickly that you barely notice! That’s why when you look at something red or blue, you instantly know what color it is without having to think too much about it.
Now let’s touch on a super interesting point: color perception. Not everyone sees colors the same way. Some people experience color blindness due to variations in their cone cells. Imagine seeing the world without being able to distinguish between greens and reds! It can be a pretty wild experience.
Also, did you know that colors can affect our feelings? Take a bright yellow—a lot of people feel happy looking at yellow because it’s often associated with sunshine and warmth. In contrast, darker colors might give off more somber vibes. This is where psychology really interacts with chromatics.
Then there’s the concept of elemental spectra. When scientists analyze materials (like gases), they can look at how those materials emit or absorb particular wavelengths of light through something called spectroscopy. For example, each element has its own unique spectral fingerprint that helps identify what it’s made of—how cool is that?
Think about stars—when astronomers use this method on star light, they can tell what elements are present in those distant suns just by analyzing their spectra! So basically through examining colors emitted or absorbed by these elements in space, we learn loads about their compositions.
In summary:
- Chromatics: The study of colors combining physics and biology.
- Light Waves: Different wavelengths correspond to different colors.
- The Retina’s Cones: Cells that help us perceive color.
- Color Perception Variability: Not everyone sees colors identically.
- Color Psychology: Colors affect emotions differently.
- Spectroscopy: Analyzing elemental spectra tells us about composition.
Color isn’t merely an aesthetic thing; it’s deeply rooted in science! Whether you’re painting a masterpiece or watching leaves change in fall, remember there’s so much happening behind those vibrant hues!
The Science of Colors: Understanding Chromatics in the Field of Physics
The science of colors, or chromatics, is like opening a window to a vibrant world. Colors aren’t just pretty; they tell us a lot about the light around us and how we perceive it. It’s interesting how what we see is all linked back to physics. Let me break it down for you.
What are Colors?
Colors come from light. When light hits an object, some wavelengths bounce off while others get absorbed. The color you see depends on the wavelengths that reach your eyes. For example, if you look at a red apple, it’s because that apple reflects red wavelengths and absorbs others. Pretty cool, right?
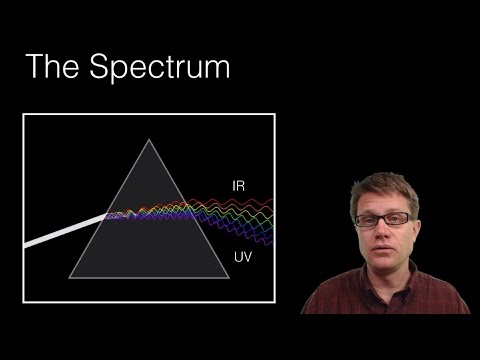
The Spectrum
Light can be split into a range of colors called the spectrum. This spectrum is made up of visible light and includes reds, greens, blues, and everything in between. You can see this when sunlight shines through a prism; it creates a rainbow effect!
- Visible Light: The part of the spectrum we can see ranges from about 380 to 750 nanometers.
- The Rainbow: Red, orange, yellow, green, blue, indigo, violet—these are the main colors in that spectrum.
Wavelengths and Frequency
Now here’s where it gets science-y: every color has its own wavelength and frequency. Wavelength is the distance between two peaks of light waves. You can think of it like waves in the ocean — closer together means higher frequency and shorter wavelengths.
- Red: Has longer wavelengths (around 620-750 nm).
- Blue: Shorter wavelengths (around 450-495 nm).
These differences cause our eyes to perceive different colors as distinct hues.
The Human Eye
Our eyes are amazing! They have cells called cones that let us see color. There are three types of cones sensitive to different ranges of wavelengths:
- L-cones: Sensitive to long wavelengths (reds).
- M-cones: Sensitive to medium wavelengths (greens).
- S-cones: Sensitive to short wavelengths (blues).
When light hits these cones, they send signals to our brain that help us understand the color we’re seeing.
The Role of Physics in Color Perception
Physics plays a huge role in how we perceive colors through phenomena like refraction and diffraction. Refraction occurs when light bends as it passes through different mediums — think about how a straw looks bent in a glass of water! Diffraction refers to how light spreads out when passing around obstacles or through narrow openings.
Both effects contribute to why we see colors differently based on context:
- Differing backgrounds: A red dress appears brighter against white than against black.
- Luminosity changes: Daylight makes colors pop more than artificial lighting.
In essence—and this might sound cheesy—color is all about interaction: how light interacts with objects and how our eyes interpret those interactions.
Anecdote Time!
I remember staring at a sunset one evening; the sky turned vibrant oranges and purples all at once! It was breathtaking but also struck me as profound because every hue was due to the scattering of sunlight by particles in the atmosphere—a little bit like magic!
So there you have it—the colorful world offered by chromatics isn’t just beautiful; it’s grounded deeply in physics principles that shape our universe! Color isn’t just what meets the eye; it’s an experience crafted by nature’s own logic.
Exploring Isaac Newton’s Colour Theory: A Comprehensive PDF Guide for Science Enthusiasts
The colorful journey into Isaac Newton’s color theory is like a treasure map! You see, Newton wasn’t just a brilliant guy who figured out gravity; he also opened the door to understanding how we perceive light and color. It all started back in the 1660s when he shone sunlight through a prism. Can you imagine that moment? Light splitting into a rainbow of colors right before his eyes!
Newton’s Color Wheel became super famous because he proposed that white light is actually made up of different colors. He divided these colors into seven categories: red, orange, yellow, green, blue, indigo, and violet. This was groundbreaking stuff! So next time you see a rainbow or play with colored lights, think about how Newton made this all possible.
Now, let’s get into the nitty-gritty of his findings. When Newton observed light through that prism, it wasn’t just magic; it was science doing its thing! He noted how each color bends at different angles when passing through the prism. This bending is called refraction. For example:
- Red light bends less than blue light. It’s all about their wavelengths!
- The shorter the wavelength (like blue), the more it bends.
- Longer wavelengths (like red) travel straighter.
Newton also connected colors to musical notes—a pretty cool way to think about them! He associated each color with specific notes in music, which makes you feel like there’s harmony in nature itself. Yeah, I know—it sounds poetic!
Furthermore, he introduced the concept of complementary colors. These are pairs of colors that cancel each other out when combined. Think about red and green or blue and orange—it’s like they’re frenemies on the color wheel! Have you ever mixed paints? You might have noticed how mixing certain colors can produce unexpected shades.
Newton’s experiments didn’t stop there; he explored color mixing. He discovered that mixing red and yellow gives you orange—simple enough—but this laid the groundwork for what we now understand as primary and secondary colors.
Another fascinating aspect is how our perception of color isn’t just physical but also psychological. Different cultures see and interpret colors differently! For instance:
- Red can symbolize love or danger.
- Blue, often seen as calming or associated with sadness.
It makes sense why artists pay such close attention to color theory—they’re tapping into both science and emotion!
In modern times, we still use Newton’s groundwork for things like photography, television screens, and even painting techniques. Although technology has expanded on his ideas with stuff like RGB (red-green-blue) systems used in digital screens today.
So there you have it! Newton wasn’t just crunching numbers under an apple tree; he was defining our understanding of color that still influences science and art today. Next time you’re outside enjoying a sunset or playing with lights at home, picture good ol’ Isaac looking over your shoulder with a proud smile because without him exploring those vibrant hues could’ve taken much longer!
So, let’s chat about something that might seem a bit academic at first but is actually really cool: elemental spectra. It’s basically how we can see the colors of elements, and it all comes down to light and atoms.
You know, I remember this one time in school, we had this science project where we got to use a spectroscope. It was like this magic device that showed us the light from different elements. When we looked through it, the colors danced before our eyes! Reds, blues, greens—each element had its own little rainbow. It felt like peering into another world, where each color told its own story.
So here’s the scoop: every element has a unique fingerprint in the form of its spectral lines. When you heat something up—like a piece of metal—it gets excited (not like at a party but more like on a molecular level). That excitement causes them to emit light at certain wavelengths. And those wavelengths? They’re what we see as color.
Think about it: when you shine white light through a prism, you get that beautiful spectrum of colors—red, orange, yellow, green, blue, indigo, violet… You follow me? Each element has its version of that spectrum when you analyze it with special equipment. Oxygen might give off colors different from sodium or copper. It’s like each one has its own playlist!
But what’s really mind-blowing is how scientists use these spectra to learn about stars or other distant celestial bodies! Seriously! By analyzing starlight through telescopes equipped with spectroscopes (there’s that word again), they can identify what elements are present in stars millions of light-years away. Can you imagine? Just by looking at some colors!
This colorful world isn’t just limited to science labs or outer space either; it surrounds us every day! Think about fireworks bursting in the sky or even pigments in paints and dyes—they all stem from understanding these elemental spectra.
And yes, while the science behind it can be complex—with quantum mechanics and all—the beauty lies in how each color connects us back to those core building blocks of nature: atoms and energy.
So next time you see a rainbow or even just the cool glow of a neon sign at night, think about all those electrons hopping around and creating their magical dance of colors! There’s something comforting knowing that behind every vibrant hue is an intricate story written in wavelengths; it reminds us how interconnected everything really is. Pretty amazing when you stop to think about it!